 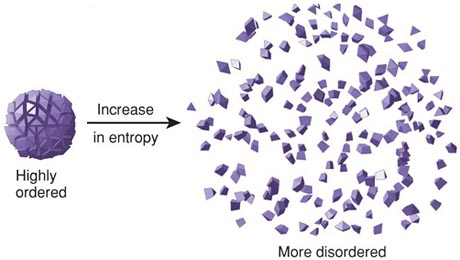
Thus although the free energy always falls when a gas expands or a chemical reaction takes place spontaneously, there need be no compensating increase in energy anywhere else. Free Energy is not energy: A much more serious difficulty with the Gibbs function, particularly in the context of chemistry, is that although G has the units of energy (joules, or in its intensive form, J mol –1), it lacks one of the most important attributes of energy in that it is not conserved.This is most commonly in the form of electrical work (moving electric charge through a potential difference), but other forms of work (osmotic work, increase in surface area) are also possible. Chemists make determinations on which chemical reactions are spontaneous and which ones are not. Calculate the entropy change for 1.0 mole of ice melting to form liquid at 273 K. The enthalpy of fusion for water is 6.01 kJ/mol. Newman b : a process of degradation or running down or a trend to disorder The deterioration of copy editing and proof-reading, incidentally, is a token of the cultural entropy that has overtaken us in the postwar years. The entropy change for a phase change at constant pressure is given by. By "useful", we mean work other than that which is associated with the expansion of the system. Entropy changes are an order of magnitude smaller than enthalpy changes, so entropy is measured in joules rather than kilojoules. Chemistry: Chemistry is the systematic study of how chemical elements combine with each other to form compounds. Entropy is the general trend of the universe toward death and disorder. Entropy is the number of possible ways quanta (packets of energy) can be distributed between the particles in a system. At this level, in the past, we have usually just described entropy as a measure of the amount of disorder in a system. The “free” part of the older name reflects the steam-engine origins of thermodynamics with its interest in converting heat into work: ΔG is the maximum amount of energy which can be “freed” from the system to perform useful work. 
Free Energy is not necessarily "free": The appellation “free energy” for G has led to so much confusion that many scientists now refer to it simply as the Gibbs energy.Electrochemical batteries are currently a major enabler in the energy transition however, batteries involve redox reactions during charging/discharging, thereby limiting their primary use in applications with relatively steady and ‘slow’ energy output (for example, portable electronics and electric vehicles).\) There is broad demand to accelerate the transition to more efficient, less polluting, and renewable energy sources such as solar and wind energy, but these sources often cannot produce energy all the time. Due to the chaotic structure of amorphous materials, it is challenging to identify defects in metallic glasses. For different substances in the same physical state at a given temperature. Defining Entropy The entropy (S) of a given system is the number of possible arrangements of the particles and their energy in a given system When a system. Recovering the stored energy requires breaking and forming new chemical bonds (typically via combustion) and, while this can be effective, it can also produce pollution that contributes to climate change. For a given substance, its entropy depends on its phase with Ssolid < Sliquid < Sgas. The challenge of storing energy is not new, and for generations, we have relied on chemical approaches (that is, energy is stored in the arrangement of atoms in molecules, as in carbon-based fuels) to this end. We face a colossal energy storage problem with analysts predicting a more than 120-times increase in global energy storage needs by 2040 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
